What Do the Four Quantum Numbers Describe About an Electron
The principle quantum number n describes the energy and distance from the nucleus and represents the shell. So pincipal quantum number is 3.
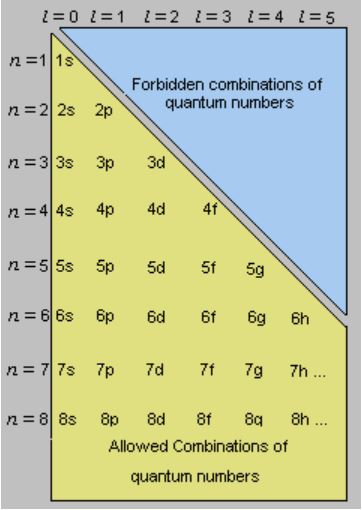
Quantum Numbers Principal Azimuthal Magnetic And Spin Definition Detailed Explanation Videos And Faqs Of Quantum Numbers
Principal Quantum Number n.

. Tell what the 4 quantum numbers represent. To completely describe an electron in an atom four quantum numbers are needed. _D_ Defines the electrons spin.
Diagram of the nuclear composition and electron configuration of an atom of phosphorus-31 atomic number. To completely describe an electron in an atom four quantum numbers are needed. Energy n angular momentum ℓ magnetic moment mℓ and spin ms.
The principle quantum number is the energy level of an electron. To completely describe an electron in an atom four quantum numbers are needed. The Four Quantum Numbers.
The s-electron magnetic quantum numbers are 0 spin quantum numbers are. The angular momentum number is the shape of the orbital holding the electron. They are the principal quantum number n the angular momentum quantum number l the.
Where value of n is 12 n it must be positive integer. Electron position within the sublevel. Energy n angular momentum ℓ magnetic moment mℓ and spin ms.
1Principal quantum number nThis quantum number is the one on which the energy of an electron in an atom principally depends. The first quantum number describes the electron shell or energy level of an atom. It determine the energy of electron in particular shell.
Not Just for Electrons While quantum numbers are commonly used to describe electrons they may be used to describe the nucleons protons and neutrons of an atom or elementary particles. The first quantum number describes the electron shell or energy level of an atom. Energy n angular momentum ℓ magnetic moment m ℓ and spin m s.
L cursive ell the shape of the electron orbital. The first quantum number describes the electron shell or energy level of an atom. This is the same as the energy level of the electron.
It can have ant positive value 123 and so onThe energy of an electron in an atom. The magnetic quantum number is the position of an orbital holding an electron. L the angular momentum quantum number tells us about the shape and the subshell which the pertaining.
Four quantum numbers are used to describe electrons. The angular momentum number is the shape of the orbital holding the electron. Energy n angular momentum ℓ magnetic moment mℓ and spin ms.
The allowed values and general meaning of each of the four quantum number of an electron numbers of an electron in an atom are as follows. 4 rows To completely describe an electron in an atom four quantum numbers are needed. The s-electrons have orbital quantum number 0 the p-electrons 1.
The first quantum number describes the electron shell or energy level of an atom. Angular Quantum Number symbol. N the size of the electron orbital.
Find Carbon on the Periodic Table. Thus for K it is View the full answer. It is the first number and identifies the principle energy level - it indicates the average or.
Spin of the electrons. What are the 4 quantum numbers describe each. Identify the 4 quantum numbers for the last valence electron in a Carbon atom.
Part B _C_ Defines the electrons orbital. Also the greater the energy level. Four quantum numbers are used to describe electrons.
Part A The four quantum numbers tell you the location and energy of an electron. The value of n ranges from 1 to the shell containing the outermost electron of that atom. The electrons in an atom are identified by aset of four quantum numbers ie.
Quantum numbers give the orbital shape the size and the orientation and spin value of the electron in question n the principle quantum number tells about the sizeshell of the orbital and about the relative energy of the orbital which the electron is in. The four quantum numbers used to describe the electrons are n2 ℓ1 m1 0 or -1 and s12 the electrons have parallel spins. It tells in which shell the electron is located.
Each electron has four set of quantum number which describe the exact position of electron in electron cloud 1-principal quantum number which is designated by n. Periodic Table for Example Problems. What is the principle quantum number n.
Thus it takes three quantum numbers to define an orbital but four quantum numbers to identify one of the electrons that can occupy the orbital. Likewise people ask what do the 4 quantum numbers mean. Principle Quantum Number symbol.
Click to see full answer. It measure distance of electron from nucleus of atom. No two electrons will have same set of its four quantum numbers.
Its electron configuration is 3s2 3p2. Energy n angular momentum ℓ magnetic moment mℓ and spin ms. The spin quantum number is the spin of an electron.
_A_ Defines the electrons energy level. The first quantum number describes the electron shell or energy level of an atom. To completely describe an electron in an atom four quantum numbers are needed.
Silicons valence electrons are in shell 3. Principal energy level of the electron. One of the electrons in an orbital is arbitrarily assigned an s quantum number of 12 the other is assigned an s quantum number of -12.
The bigger the orbital the greater the distance of the electron from the atomic nucleus. _B_ Defines the electrons sublevel. The four quantum numbers are the principle quantum number n the angular momentum quantum number l the magnetic quantum number m_l and the electron spin quantum number m_s.
To completely describe an electron in an atom four quantum numbers are needed. The four quantum numbers used to describe the electrons are n2 ℓ1 m1 0 or -1 and s12 the electrons have parallel spins.

Quantum Numbers Principal Azimuthal Magnetic Videos And Examples

Hydrogen Atom And Quantum Numbers Youtube

Question Video Determining The Quantum Numbers That Represent An Electron In An Atom Nagwa

Quantum Numbers Atomic Orbitals And Electron Configurations

Quantum Numbers Video Quantum Physics Khan Academy

Question Video Ranking Three Electrons From Lowest To Highest Energy Given The Quantum Numbers Of Each Nagwa

Quantum Numbers Orbital Shapes Chemtalk

The Four Quantum Number Of The Valence Electron Of Potassium Are Youtube
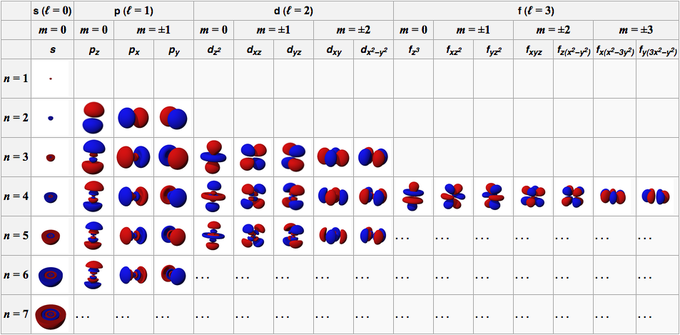
Quantum Numbers Introduction To Chemistry

How To Determine The 4 Quantum Numbers From An Element Or A Valence Electron Youtube
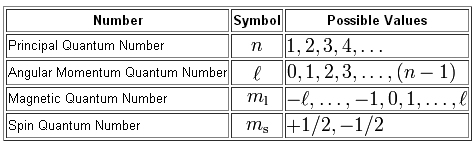
Quantum Numbers Introduction To Chemistry

Quantum Numbers Explained Chemistry Help Chemistry Lessons High School Chemistry
Can You Explain The Significance Of 4 Quantum Numbers Quora
What Are The Four Quantum Numbers Corresponding To The 15th Electron Of Phosphorus Quora
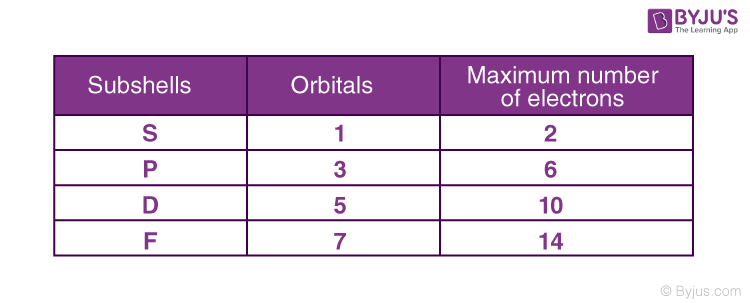
Magnetic Quantum Number Definition Schrodinger Equation

Quantum Numbers The Easy Way Youtube

Orbitals And Quantum Numbers Ppt Download
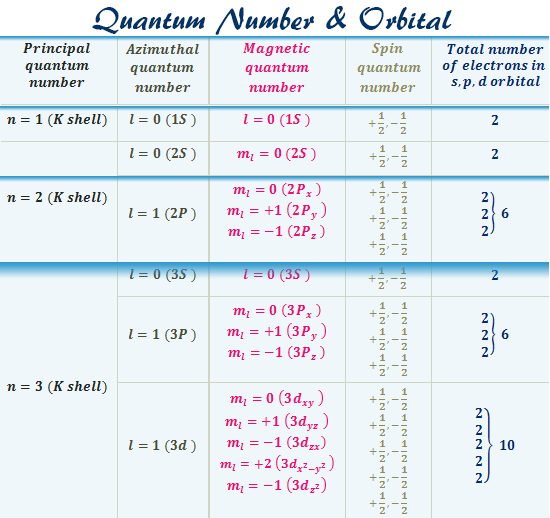
Quantum Number Orbital Definition Formula Diagram Shape

Quantum Numbers For The First Four Shells Video Khan Academy

Comments
Post a Comment